Introduction : NCAM1 (CD56) is a cellular adhesion molecule highly expressed on natural killer (NK) cells and other immune cells. NCAM1 is aberrantly expressed in 20% of patients with acute myeloid leukemia (AML) and is associated with resistance to chemotherapy, upregulation of the MAPK pathway, and poor prognosis. The significance of NCAM1 in the setting of venetoclax (VEN) therapy has not been explored. We set out to determine the functional and prognostic role of CD56 in vitro and in newly diagnosed (ND) patients receiving VEN-based therapy.
Methods : OCI-AML2, KG-1, and MOLM-14 cell lines were purchased from ATCC and maintained in RPMI, 10% or 20% FBS, L-Glutamine, and penicillin/streptomycin. Ex vivo drug testing on cell viability was performed using an MTS assay. Briefly, AML cells were plated for 72 hours in replicates of six at 1,250 cells/well in R20 and titrated using a log scale over 7 doses (0-1000 nM) of VEN. After 3 days of culture at 37 °C in 5% CO2, MTS reagent was added, optical density was measured at 490 nm, and raw absorbance values were adjusted to a positive control and (normalized to untreated wells to produce cell viability estimates). VEN-resistant (VR) OCI-AML2 cells were generated by continuous incubation with escalating doses of VEN. Viability was monitored by Guava EasyCyte. Cells maintained resistant by co-culture with 1 µM of VEN twice weekly. Flow for NCAM1 was performed with commercially available antibodies on parental and VR cells. NCAM1 expression on parental vs. VR OCI-AML2 cells was compared with student's T test. Inactivation of NCAM1 in OCI-AML2 cells was carried out by CRISPR/Cas9 using sgRNAs cloned into plentiCRISPRv2 (Addgene, #52961). AUC was derived from 4-parameter logistic regression curves fit to normalized cell viability values for the 7 tested drug doses. Clinical outcomes including overall survival (OS) were assessed in a retrospective cohort of ND AML patients treated with a hypomethylating agent (HMA)+VEN at Oregon Health & Science University (OHSU). CD56 positivity on AML blasts was based on surface immunophenotype determined by multiparameter flow cytometry. OS was measured from AML diagnosis (with time from diagnosis to VEN start as left-truncation), estimated by Kaplan-Meier, compared with the log-rank test, and modeled with Cox regression to estimate unadjusted and adjusted hazard ratios (HRs) for CD56.
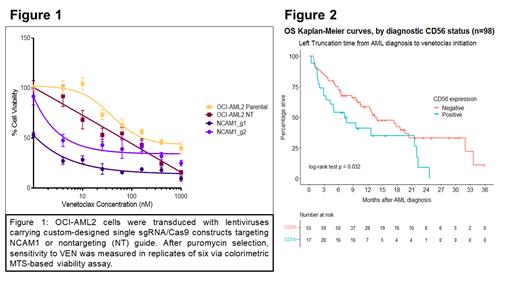
Results : Across the 3 AML cell lines evaluated, higher baseline NCAM1 expression levels correlated with increased ex vivo resistance to VEN. The MOLM14 cell line was most resistant with highest median NCAM1 expression (90%), followed by OCI-AML2 with median NCAM1 expression (55%) and then KG-1 with lowest NCAM1 expression (3%). VR OCI-AML2 cells had higher expression of NCAM1 compared to parental OCI-AML2 cells (80% expression vs 55%; p=0.0003). CRISPR knockout of NCAM1 sensitized parental OCI-AML2 cells to VENcompared to cells containing a non-targeting control guide (Fig 1). To support our pre-clinical findings, we retrospectively identified N=98 ND AML patients treated with HMA+VEN at OHSU from 12/2018 to 12/2022. Median follow-up was 16 months and 60% of patients died. Median age at diagnosis was 73 (range=19-87), with 39% at least 75 years old. While 65% of patients were male, 63% had de novo AML and 23% had an antecedent hematologic disorder (AHD). By European LeukemiaNet (ELN) 2022 prognostic risk classification, 58% had adverse risk (14% unknown). The most common somatic mutations were IDH1/2 (27% of patients), NRAS/KRAS (26%), FLT3 ITD or TKD (19%), NPM1 (13%), and TP53 (13%). According to diagnostic flow, 30% of patients were CD56+, 22% CD11b+, 15% CD64+, and 85% HLA-DR+. Median (range) days between diagnosis and VEN start was 13 (1-285). NCAM1 positivity significantly correlated with shorter OS in the univariable (HR=1.80 [95% CI: 1.04-3.09], p=0.032; Fig 2) and multivariable settings (HR=2.10 [95% CI: 1.17-3.75], p=0.012, N=97), with the latter model controlling for age ≥75, AHD, and CD11b.
Conclusion : NCAM1 expression is a marker of poor prognosis in ND AML patients treated with HMA+VEN. Knockout of NCAM1 in the OCI-AML2 cell line sensitized cells to VEN. Our data suggest that NCAM1 expression on AML blasts plays a biological role in promoting cellular resistance to VEN. Given emerging knowledge of VEN targeted downstream pathway associations, experiments are underway to explore the role of MAPK pathway and BCL2 family member dependence.
Disclosures
Saultz:Rigel: Other: Advisory Board; IKENA Oncology: Research Funding. Lachowiez:Rigel Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; COTA Healthcare: Consultancy. Lind:Kronos Bio: Research Funding. Leonard:Takeda: Consultancy; Kite/Gilead: Consultancy; Adaptive Biotechnologies: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel, accommodations, expenses; Pfizer: Consultancy. Maziarz:Orca Therapeutics: Research Funding; Athersys: Other: Patent holder; Kite: Consultancy; Gamida: Research Funding; AlloVir: Consultancy, Research Funding; Novartis: Consultancy, Research Funding. Traer:Incyte: Research Funding; Prelude Therapeutics: Research Funding; Schrodinger: Research Funding; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Membership on an entity's Board of Directors or advisory committees; Servier: Membership on an entity's Board of Directors or advisory committees; Astra-Zeneca: Research Funding; Rigel: Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees. Swords:Kronos Bio: Research Funding. Braun:Blueprint Medicines: Consultancy, Research Funding; Gilead Sciences: Research Funding; Novartis: Consultancy; Oryzon Genomics: Other: Institutional PI (FRIDA trial); AstraZeneca: Research Funding. Tognon:Notable Labs: Research Funding. Tyner:Schrodinger: Research Funding; Acerta: Research Funding; Constellation: Research Funding; Tolero: Research Funding; Recludix Pharma: Membership on an entity's Board of Directors or advisory committees; Aptose: Research Funding; AstraZeneca: Research Funding; Kronos: Research Funding; Meryx: Research Funding; Petra: Research Funding; Incyte: Research Funding; Genentech: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal